Kinetic Study of Congo-Red Photo-Catalytic Degradation in Aqueous Media Using Zinc Oxide as Photo Catalyst Under Led Light
Abstract
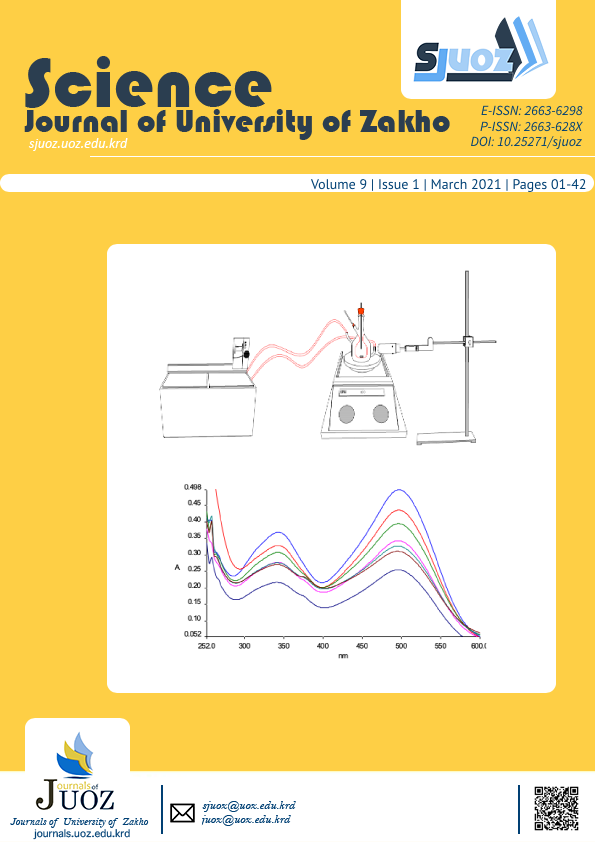
The kinetic of photo catalytic degradation of Congo red dye using semiconductor in aqueous solution of ZnO has been studied. All photochemical experiments have been carried out in quartz photo cell 25ml capacity and the solvent used for all experiment was distilled water (D. W). The influence of temperature has been investigated as well. Spectrophotometric method was utilized for this work. Degradation kinetic has been done for absorption peak of Congo red at 497 nm and 344 nm. Different method was applied for this purpose, and the results show that the degradation of Congo red was first order at 497nm and zero order at 344 nm. The degradation of CR dye were increased by increasing temperature from 20 oC to 40 oC and then degreased at 50 oC and this is due to desorption that occur at the semiconductor surface while; in case of 10 oC the rate constant was higher than 20 oC. However, when changing the concentration the rate constant dose not changed regularly, this perhaps due to the fact that it does not follow the same order throughout the degradation process, and it does not obey Arrhenius law of activation energy.
Full text article
References
Authors
Copyright (c) 2021 Shinwar A. Idrees, Raad N. Salih, Khalat Bashir, Aram A. Hamasaeed

This work is licensed under a Creative Commons Attribution 4.0 International License.
Authors who publish with this journal agree to the following terms:
- Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution License [CC BY-NC-SA 4.0] that allows others to share the work with an acknowledgment of the work's authorship and initial publication in this journal.
- Authors are able to enter into separate, additional contractual arrangements for the non-exclusive distribution of the journal's published version of the work, with an acknowledgment of its initial publication in this journal.
- Authors are permitted and encouraged to post their work online.